Tech
At it’s heart, the automated SISCAPA workflow consists of specific immuno-affinity capture and enrichment of selected target peptides and their corresponding stable isotope-labeled internal standards from a proteolytic sample digest.
Digestion of sample proteins eliminates common assay interferences caused by protein:protein interactions, while specific enrichment of selected peptides eliminates matrix effects and allows collection of target from large (or small) volumes of sample.
Leveraging the absolute structural specificity available with MS detection, which removes the risk of measuring the wrong protein or the wrong form of a protein, SISCAPA enrichment improves MS assay performance in three specific ways:
- Sensitivity: Low abundance peptides can be recovered with high efficiency (e.g., >90%) from any volume of input sample (e.g., a digest of 400uL of plasma), so that sensitivity is limited only by MS performance.
- Throughput: By eliminating >99% of matrix peptides during enrichment, the need for lengthy LC separation prior to MS detection can be reduced, and in many cases eliminated (e.g., using RapidFire, Echo-MS, or MALDI).
- Dynamic Range: By capturing all of a low abundance peptide and a tiny fraction of a high abundance peptide (along with their respective IS forms), peptides differing in abundance by >1010-fold can be measured accurately in the same multiplex panel.
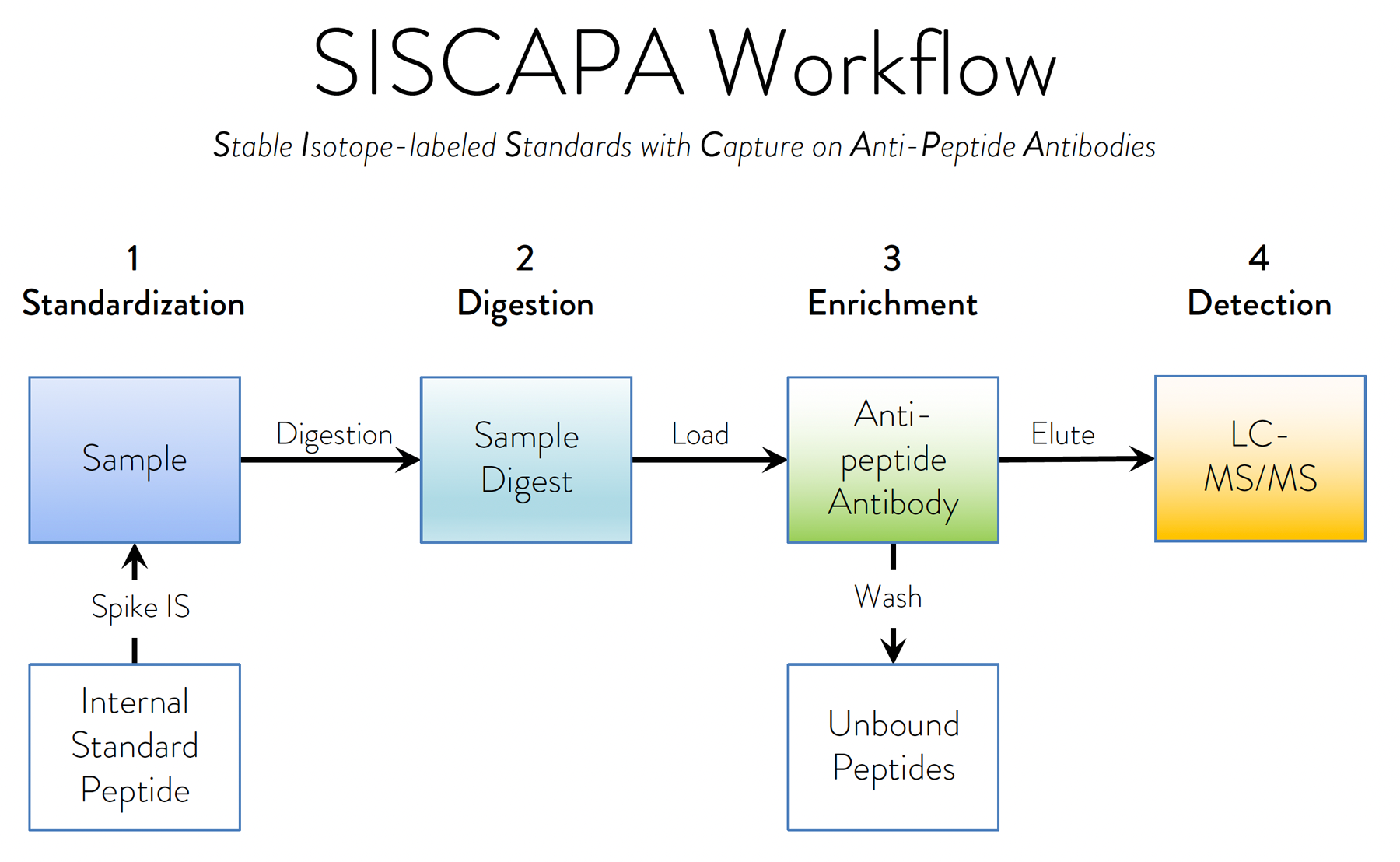
To achieve these improvements, we develop very high affinity (sub-nanomolar), low off-rate monoclonal anti-peptide antibodies to selected proteolytic peptide(s) derived from a target protein. Our extensive experience managing the required reagent generation pipeline, together with our ability to optimize SISCAPA assay performance in fully automated liquid handling platforms, enables us to provide solutions to a broad range of protein measurement challenges.
Traditional methods of protein measurement, including immunoassays or other LBA formats, can be limited by a range of specificity issues: unexpected cross-reactivity with related molecules, interferences in complex sample matrices, inability to differentiate isoforms and post-translational modifications, etc.
SISCAPA is an acronym for “Stable Isotope Standards and Capture by Anti-Peptide Antibodies”, sometimes referred to as immuno-affinity-MS.
Features
Ultra-specific
Peptide MRM mass spectrometry detection provides absolute structural specificity using genome-defined proteotypic peptides.
High-sensitivity
Immuno-affinity enrichment concentrates target peptides from small or large volumes of sample digest, increasing MS sensitivity 1,000 to 10,000 -fold.
Standardized
Stable isotope-labeled peptide internal standards provide highly accurate and inherently reproducible peptide quantitation by isotope-dilution MS.
Interference-free
Initial proteolytic digestion of samples to peptides removes all protein-protein interactions, removing the source of common assay interferences.
Easily multiplexable
Assays use a common protocol and can be flexibly combined in panels since there is no cross-assay interaction.
Wide dynamic range
Panels including peptides differing in abundance by >1 million-fold can be measured at once by exploiting SISCAPA stoichiometric flattening.
Fully automated
The SISCAPA workflow is fully automated on a variety of commercially available liquid-handling platforms as a rapid, addition-only protocol using magnetic bead capture with no additional separative steps (e.g., centrifugation or SPE cleanups), making it scalable and amenable to facile implementation.
Transferable protocol
The same SISCAPA reagents and automated workflow used for pharma R&D applications are used directly in regulated CROs for clinical trial analysis and in clinical reference laboratories to generate patient test results.
